 
One last major trend of the period table deals with the metallic character of an element. It generally increases left to right and decreases top to bottom. Is added to a neutral atom to form a negative ion. Electron affinity is the amount of energy released when an electron Trends, but they are less consistent than the others. 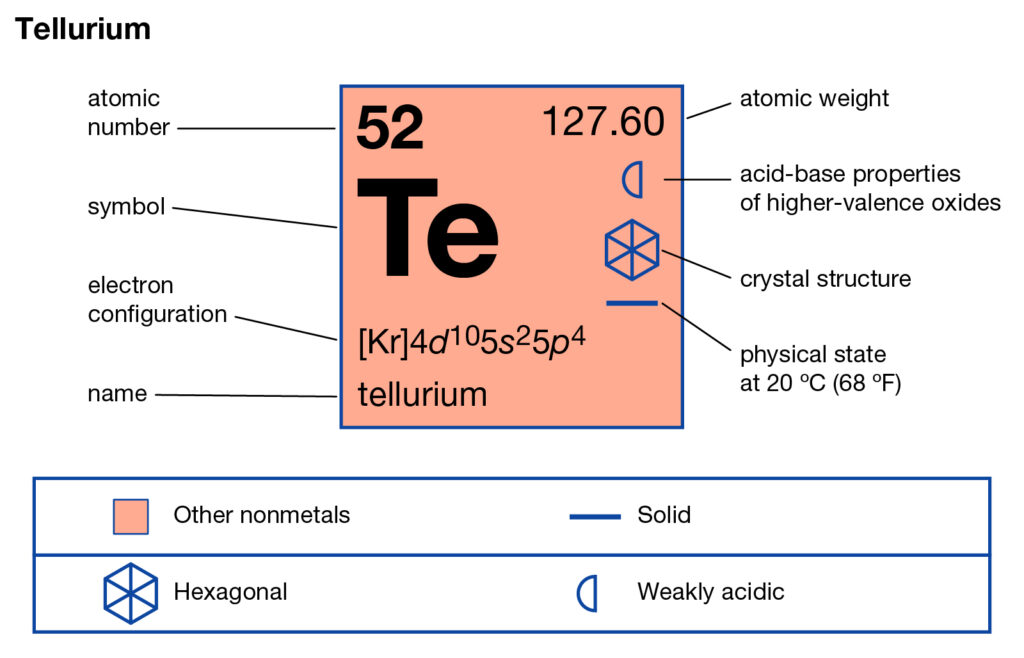
Tendency of an element to attract electrons, follows the same pattern as ionization energy. Required to remove an electron increases from left to right and decreases from top to bottom. The ionization energy of an element, or the amount of energy The atomic radius increases from top to bottom. This is because each time a proton is added the electrons are drawn closer in towards theĬenter. The size (atomic radius) of an electron decreasesįrom left to right. These deal with the size, ionization energy,Įlectronegativity, and electron affinity of an element. There are also specific trends that the periodic table follows. In order to predict the number of valence electrons that transition metals have, other methods Although helium is in group 18, it only has two valence electrons. The common group names and valance electrons areĪ few points should be noted. To tell how many valence electrons elements in certain groups have. All elements in a group have similar properties. There are ten named groups in the periodic table. Out the electron configuration of an element. Has the lanthanides and actinides usually located in two rows at the bottom of the period table. The first two groups, the p block contains the last six groups, the d block contains groups 3 to 12, and the f block Is also organized into four blocks according to the subshell in which the last electron resides. This will be discussed more in the next section. 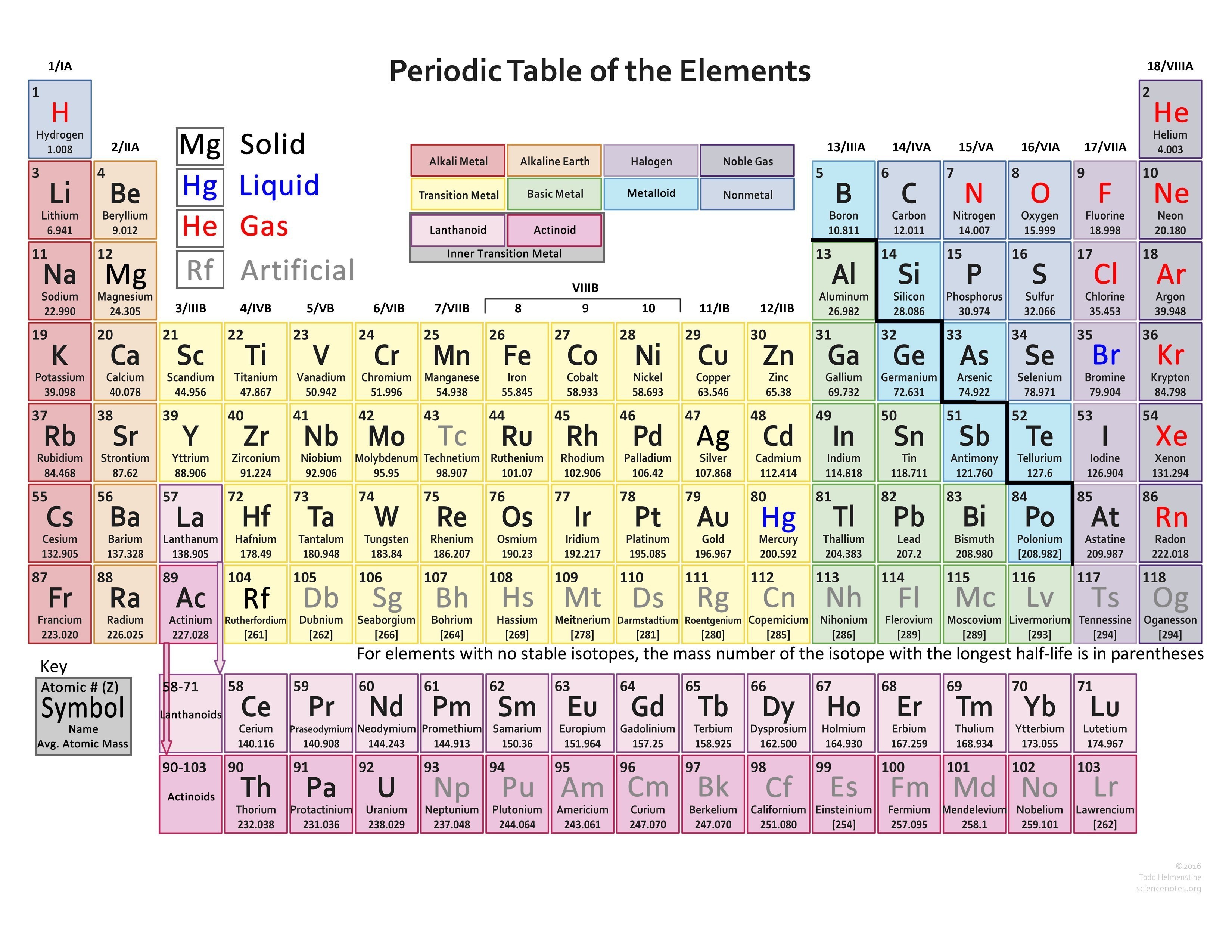
Properties an element has from its group number. We can tell the number of valence (outer) electrons as well as other useful Typically the element’s symbol, atomic number, and atomic weight are shown. The periodic table is structured in a way that is very useful for chemists. This new organization fixed all of the contradictions created by the the atomic weight organization. He did this by shooting electrons at atoms and analyzing emission x rays. The solution to this problem came in 1913 when Henry Moseley organized the elements by atomic number (the number of protons an atom has). For example, according to Mendeleev’s table, Argon should have been placed before Potassium. Mendeleev was able to predict the existence of elements before they were discovered or synthesized by looking at gaps in his table. He put the elements into a grid with increasing atomic weights and noticed the elements in each column had similar chemical properties. The first version was constructed by Dmitri Mendeleev in 1869. The periodic table is a way to organize the elements based on their similarities. The integer number in the box with the element symbol is the atomic number of the element and also the number of protons in each atom of the element. We number the elements, beginning with hydrogen, number one, in integers up to the largest number. As you read down the chart from top to bottom, a line of elements is a Group or Family. As you read across the chart from right to left, a line of elements is a Period. The Periodic Chart of the Elements is just a way to arrange the elements to show a large amount of information and organization. The Periodic Chart of Table of the Elements Overview The Periodic Chart of Table of the Elements.Explanation of States of Matter Problems.SN1SN2 – Nucleophilic Substitution Reactions.Explanation of Numbers and Math Problems – Set 3.Explanation of Numbers and Math Problems – Set 2.Explanation of Numbers and Math Problems – Set 1.Metric Prefixes and Their Origins and Use.Drawing Cyclohexane Rings – Organic Chemistry.Alkanes and Alkenes – Organic Chemistry.Light & Dark Reactions in Photosynthesis.Search Lessons Search for: Science Resources and Science Lessons – Science Help 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
